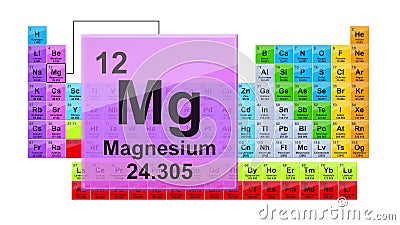 
In this video, well walk through this process for the ionic compound calcium bromide. To use this for the -ite ions, simply subtract one oxygen but keep the charge the same. Inclusion of the word 'ate' signifies that each ends with the letters a-t-e. To balance the charges, you will need two nitrate ions for every magnesium ion. Number of vowels denotes negative charge quantity. Magnesium is in Group 2 and so has 2+ charges, Mg2+. Finally, combine the two ions to form an electrically neutral compound. Number of consonants denotes number of oxygen atoms. Then, identify the anion and write down its symbol and charge. Right You have already seen the bohr model of magnesium element in the above table. Group 2A (or IIA) of the periodic table are the alkaline earth metals: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).They are harder and less reactive than the alkali metals of Group 1A. An iron ion with a charge of +8 is not very likely, therefore, the octet rule is not applicable to transition elements. To find the formula of an ionic compound, first identify the cation and write down its symbol and charge. How many shells does magnesium have It’s 3. whereas magnesium ions (Mg2+) is essential for certain enzymes, muscles. In order for an element such as iron (Fe) to achieve the same noble gas configuration of argon (Ar), it would need to lose 6 electrons in the 3 d subshell and 2 electrons in the 4s subshell. Trends in Ionic Charge: Meaning Periodic Table Ionisation Energy. This is because the transition metals have electrons in d subshell and do not follow the octet rule. Notice that there is no simple pattern for transition metal ions (or for the larger main group elements) as there is with the main group ions. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and. \) shows the characteristic charges for some of these ions. Element Magnesium (Mg), Group 2, Atomic Number 12, s-block, Mass 24.305.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
